on VALNEVA (EPA:VLA)
Pfizer and Valneva unveil promising results for their Lyme disease vaccine
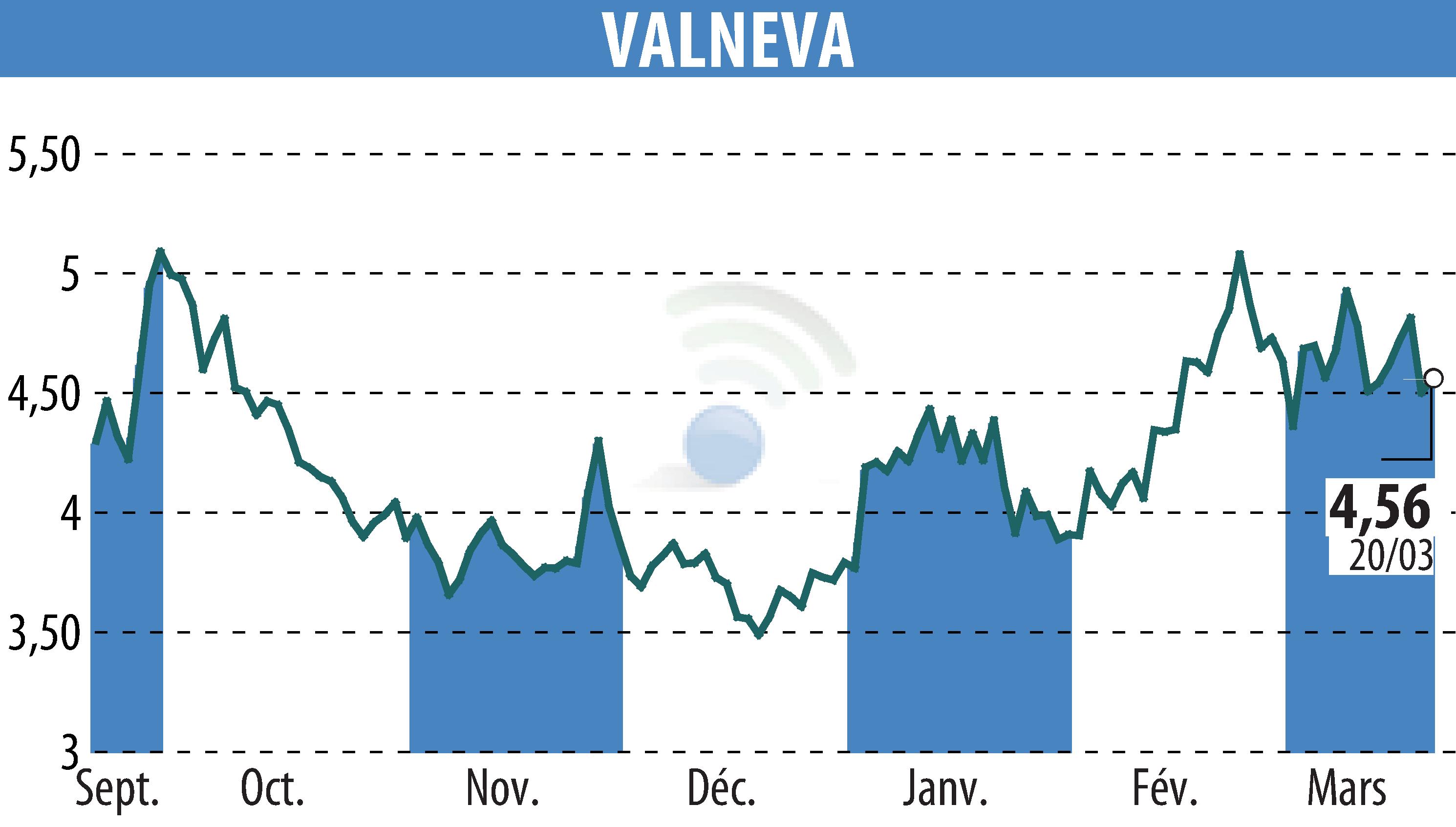
Pfizer and Valneva have announced encouraging results for their Lyme disease vaccine candidate, PF-07307405 (LB6V), from the Phase 3 VALOR trial. This candidate demonstrated efficacy of over 70% in individuals aged five years and older, with no major adverse events observed. This performance strengthens confidence in this vaccine, which aims to combat a disease for which no vaccine solution is currently available.
In pre-defined analyses, the vaccine demonstrated 74.8% efficacy after the fourth dose. Fewer cases than expected were observed, but the notable efficacy encourages Pfizer to consider submitting regulatory applications.
R. P.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all VALNEVA news
