on SANOFI-AVENTIS (EPA:SAN)
Sanofi's Vaccine Shows Superior Tolerability in Recent Study
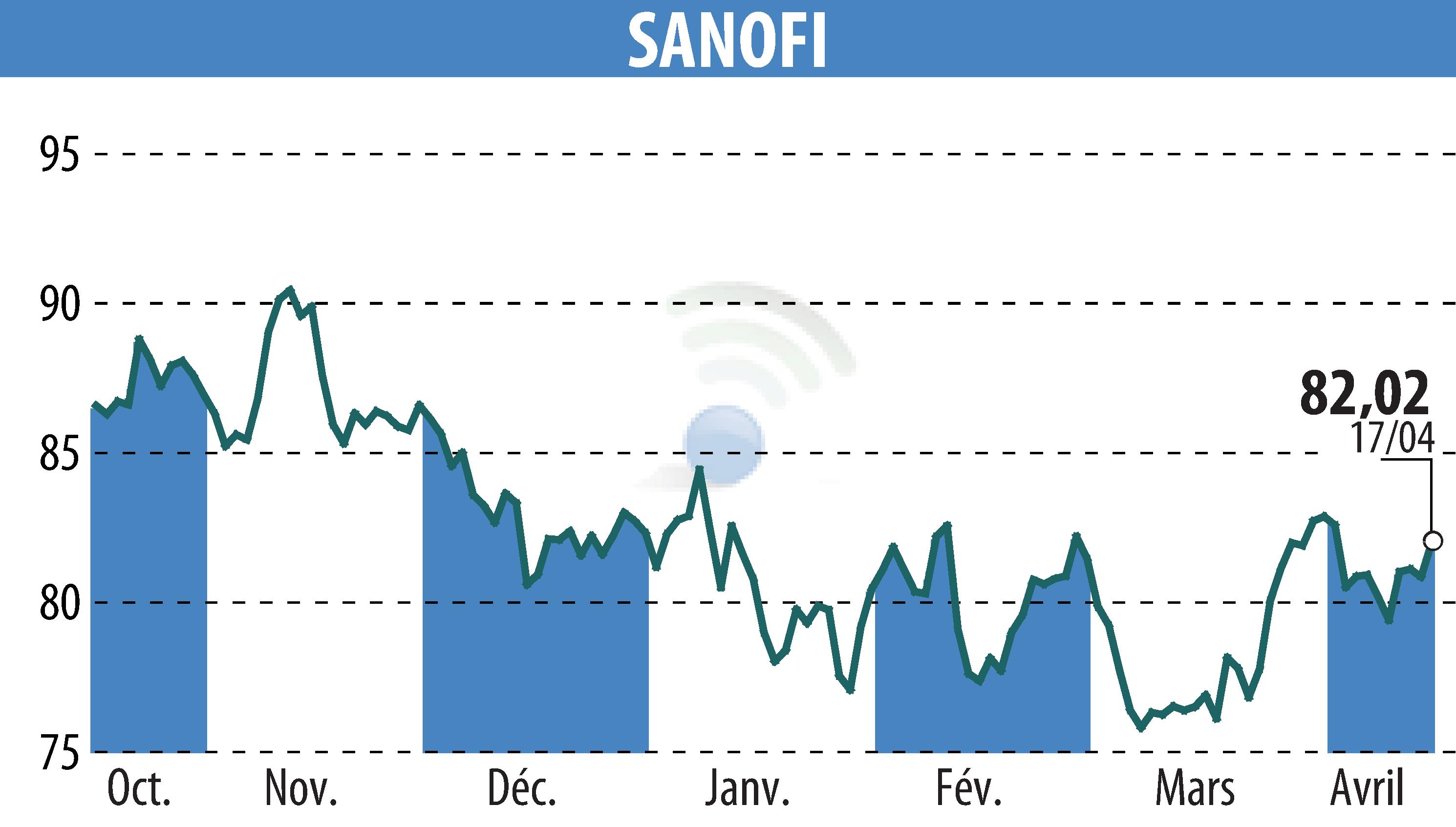
Sanofi has announced that its COVID-19 vaccine, Nuvaxovid, demonstrated better tolerability compared to Moderna's mRNA-1283, known as mNEXSPIKE, in a comprehensive phase 4 study. The findings, presented at the ESCMID Global Congress in Munich, reveal Nuvaxovid's statistically significant lower systemic reactogenicity across all pre-specified endpoints. This study enrolled 1,000 adults in the US, illustrating a key point: despite the pandemic's end, severe COVID-19 cases continue, with vaccine side effects contributing to low vaccination uptake.
The study highlighted that severe systemic symptoms occurred more than 50% less frequently with Nuvaxovid than mNEXSPIKE. Local symptoms at the injection site were also less frequent. This led to nearly double the number of Nuvaxovid recipients expressing willingness to choose the same vaccine again. Overall, fewer systemic reactions were seen with Nuvaxovid, encouraging greater vaccine confidence, potentially boosting routine immunization rates.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
