on NANOBIOTIX (EPA:NANO)
Nanobiotix Updates Protocol for Phase 3 Cancer Study
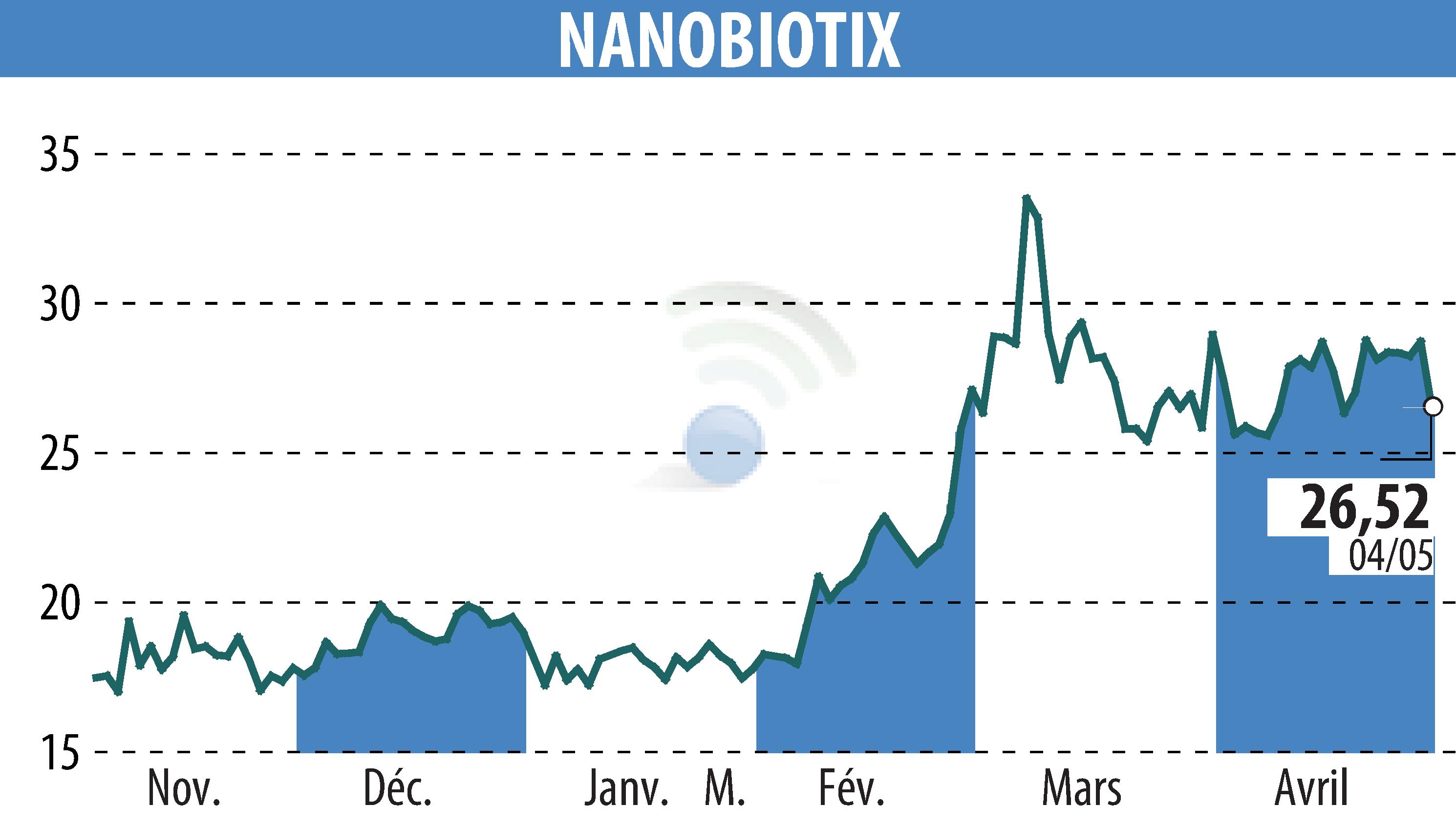
Nanobiotix, a biotechnology company, announced the FDA's acceptance of a protocol amendment for their ongoing Phase 3 NANORAY-312 study. This amendment, backed by Johnson & Johnson, removes the interim analysis. It modifies the final analysis to occur sooner with fewer events needed. The objective is to hasten the global registration path for JNJ-1900 (NBTXR3) in head and neck cancer. The modification aims to align the final analysis timing with the previously planned interim analysis, contingent on clinical events.
Nanobiotix expects the revised protocol to expedite potential revenue generation. The company is eligible for significant financial payments under a license agreement, pending milestone achievements in both head and neck and lung cancer studies.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all NANOBIOTIX news
