on NANOBIOTIX (EPA:NANO)
NANOBIOTIX Reveals Initial Data from Phase 2 Lung Cancer Trial
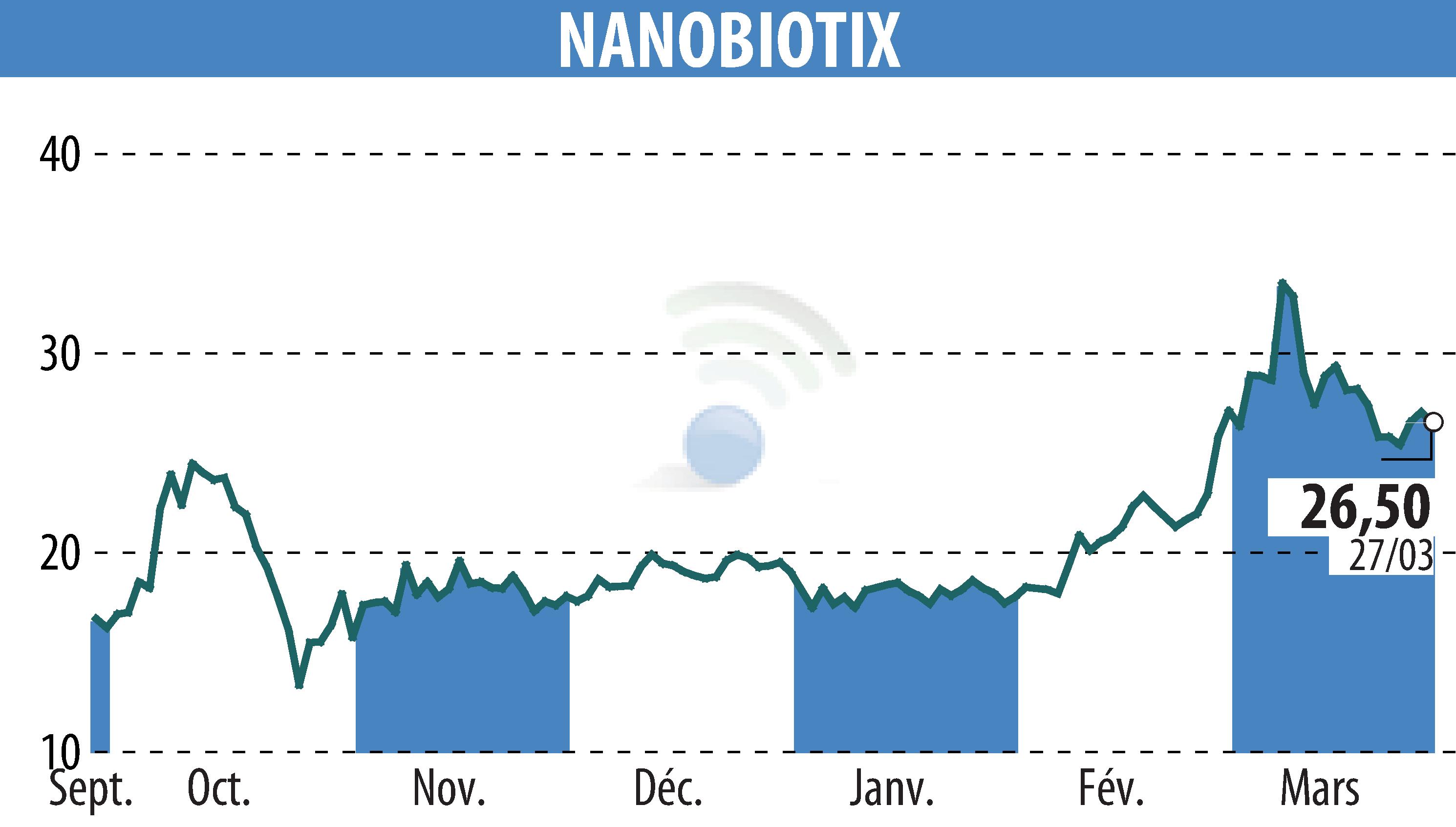
NANOBIOTIX has unveiled first data from the CONVERGE study at the 2026 European Lung Cancer Conference. The Phase 2 trial, backed by Johnson & Johnson, evaluates the nanoradioenhancer JNJ-1900 (NBTXR3) for stage 3 inoperable non-small cell lung cancer. The study highlighted an acceptable safety profile with no serious treatment-emergent adverse events. Moreover, promising initial efficacy responses were observed, with an objective response rate (ORR) of 71.4% and disease control rate (DCR) of 100% at first disease evaluation, surpassing the estimated benchmark.
JNJ-1900 (NBTXR3), designed as a novel oncology product, showed potential to transform treatment through its unique mechanism activated by radiotherapy. This promising nanotherapeutic approach reflects ongoing collaboration strategies and global co-development efforts with Janssen Pharmaceutica NV.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all NANOBIOTIX news
