on Heidelberg Pharma AG (ETR:HPHA)
Heidelberg Pharma to Receive Milestone Payment for HDP-101 Study in China
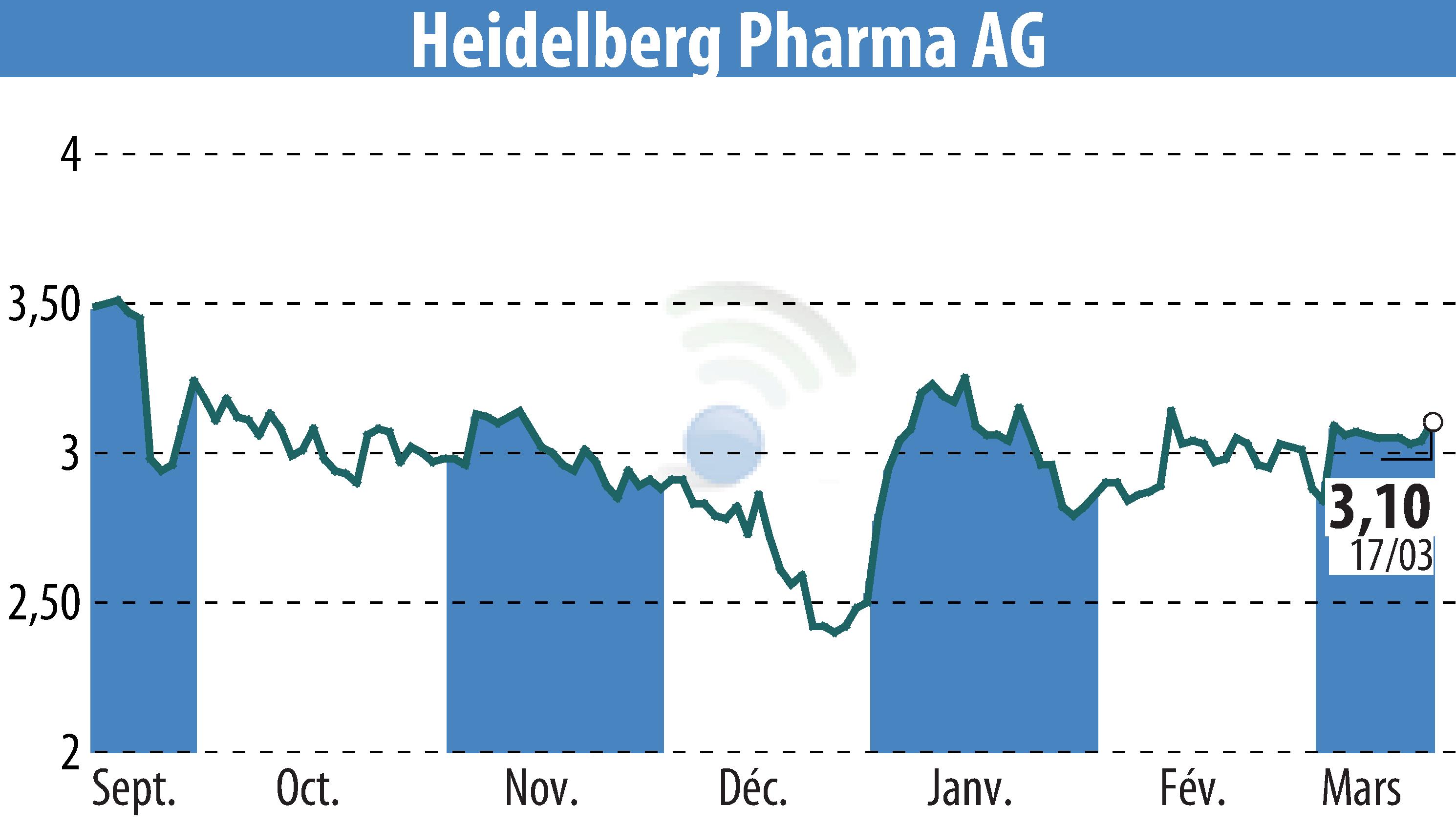
Heidelberg Pharma AG announced that it will receive a milestone payment from its partner, Huadong Medicine Co., Ltd., following the initiation of a Phase I clinical study in China with HDP-101. The study marks a significant step in evaluating the safety, tolerability, pharmacokinetics, and efficacy of pamlectabart tismanitin among patients with plasma cell disorders, including multiple myeloma.
This study, involving the first patient dose in China, aims to broaden the evaluation of the ATAC candidate's effectiveness across diverse populations. The chosen dose of 140 µg/kg was previously demonstrated to be safe. Dr. Donghzou Jeffery Liu stated that this development expands their testing landscape beyond Europe and the US to Asia, highlighting the differentiation provided by their Amanitin-based technology.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Heidelberg Pharma AG news
