on GENSIGHT BIOLOGICS S.A. (EPA:SIGHT)
GenSight Biologics: Update on Early Access Programs and the REVISED Study
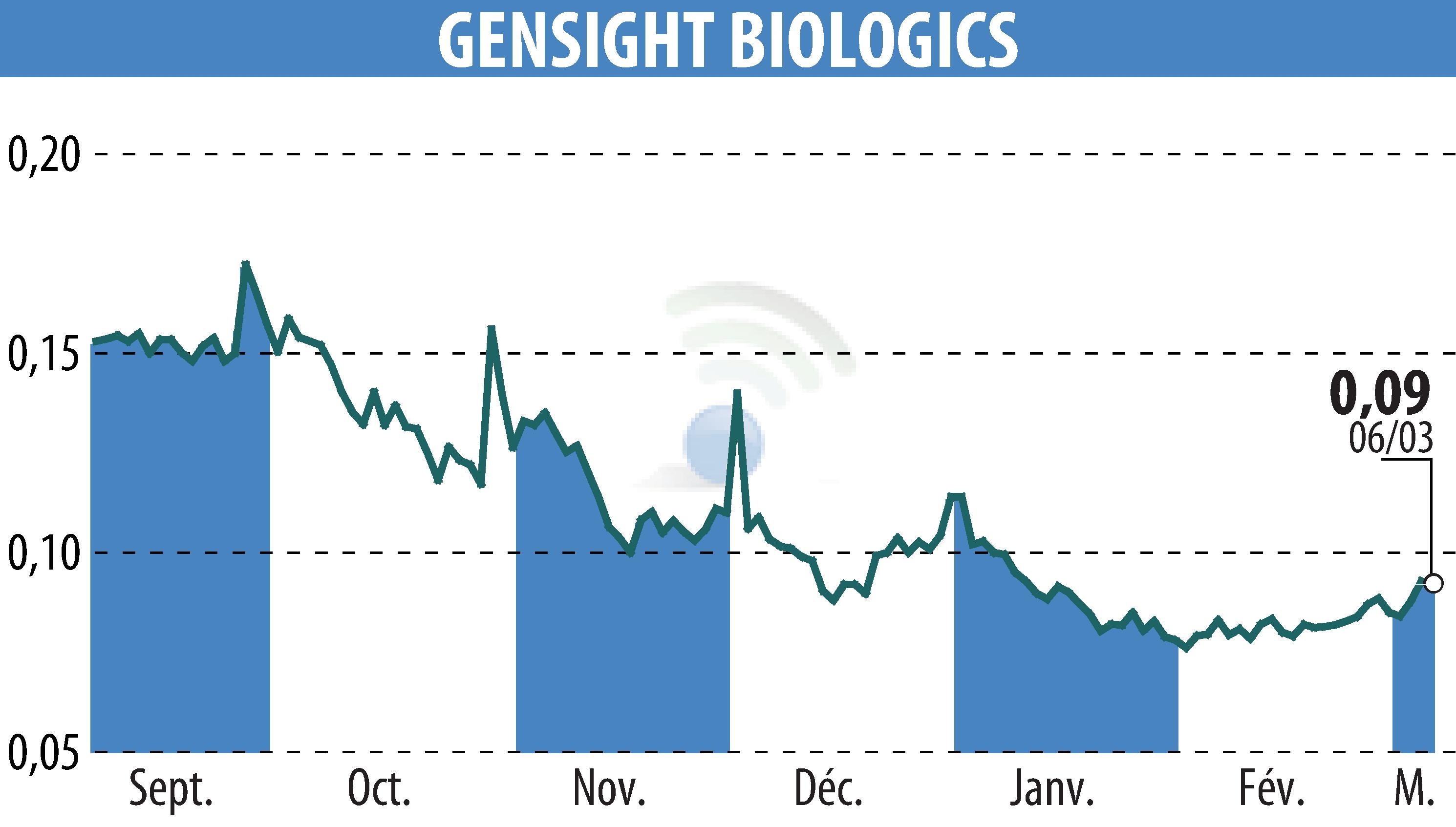
GenSight Biologics, a clinical-stage biopharmaceutical company, provides an update on its early access programs for GS010/LUMEVOQ® and the REVISE study. GS010/LUMEVOQ®, a gene therapy in development for Leber Hereditary Optic Neuropathy, continues its progress in France, Israel, and the United States.
In France, following approval of the Compassionate Access Authorization by the ANSM, treatments will begin in March at the Quinze-Vingts Hospital. The REVISE study is on schedule with one patient treated and a second to be recruited soon.
Israel has approved treatment for a second patient through the early access fee program, and in the United States, FDA approval of a second individual IND request will allow for further treatment under the "Expanded Access" program.
The expected revenues are considered sufficient to maintain the company's operations in 2026.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all GENSIGHT BIOLOGICS S.A. news
