on Eckert & Ziegler Strahlen- Und Medizintechnik AG (ETR:EUZ)
Eckert & Ziegler Gains MDR Certification for Critical Eye Cancer Treatment
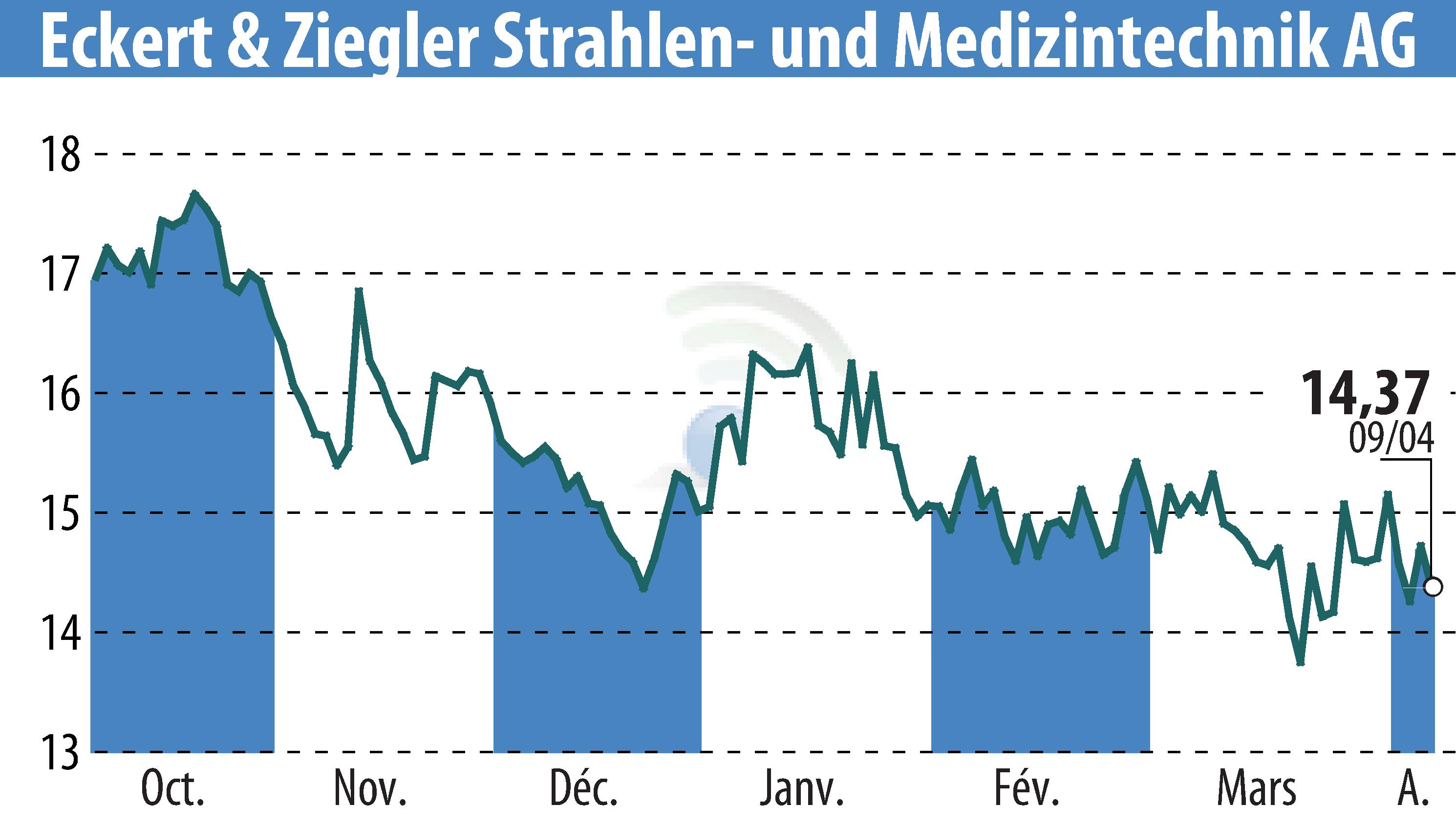
Eckert & Ziegler BEBIG GmbH, a subsidiary of Eckert & Ziegler SE, has received MDR certification for its Ru-106 Eye Applicators, ensuring continued patient access to critical eye cancer treatments. As the sole global supplier, this certification addresses potential treatment shortages.
The Medical Device Regulation (MDR) is an EU directive aimed at enhancing medical device quality and patient safety. With this certification, Eckert & Ziegler guarantees the long-term availability of its products in the EU, maintaining a product legacy important to its growth.
Ru-106 plaques are used in ophthalmic brachytherapy, treating uveal melanoma and retinoblastoma by suturing them to the eye wall to deliver radiation. The treatment aids in preserving patients' vision and quality of life.
R. E.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all Eckert & Ziegler Strahlen- Und Medizintechnik AG news
