on BIOMERIEUX (EPA:BIM)
BioMérieux Secures IVDR CE-Mark for New Diagnostic Panels
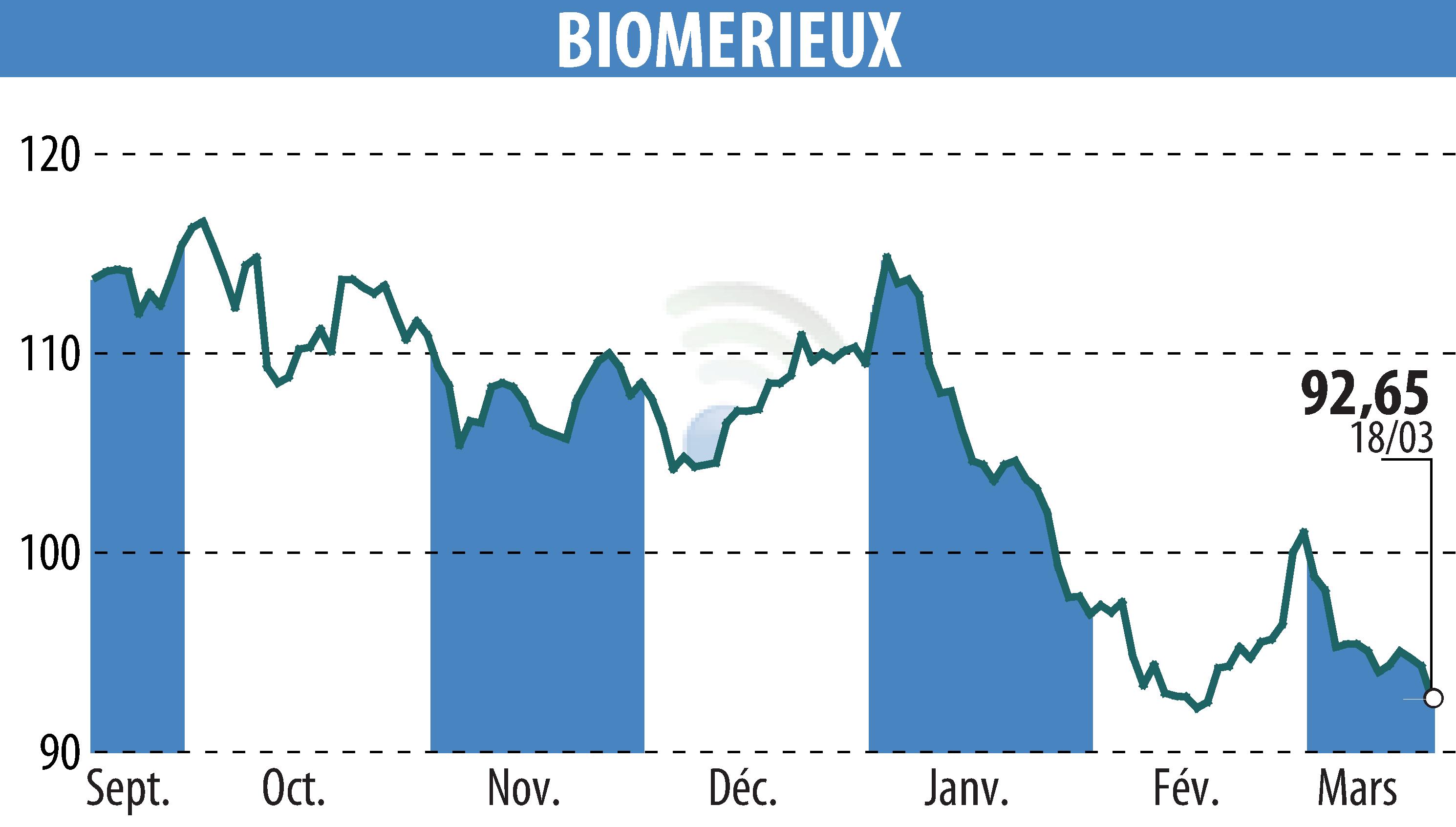
bioMérieux has received IVDR CE-marking for its BIOFIRE® SPOTFIRE® Respiratory/Sore Throat plus (R/STplus) Panels, enhancing rapid diagnostic capabilities across Europe. These PCR tests, designed for near-patient testing, can swiftly identify respiratory and sore throat pathogens. The tests are compatible with the compact BIOFIRE® SPOTFIRE® System, offering results within 15 minutes, and provide flexibility in sample collection methods.
The panels aim to tackle global health challenges by facilitating the quick identification of respiratory pathogens, which aids in appropriate treatment and reduces antibiotic misuse. The R/STplus Panel detects 15 pathogens, while the Mini version identifies up to six, including key viruses like SARS-CoV-2. These panels will be available in Europe by the second quarter of 2026, with wider availability subject to regulations.
R. H.
Copyright © 2026 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all BIOMERIEUX news
